Predictable Performance. Repeatable Results.
Automation is where process performance becomes predictable and repeatable. ABEC designs control solutions that reduce variability, improve visibility, and support reliable outcomes—from development through commercial manufacturing—across standalone skids or full facility environments.
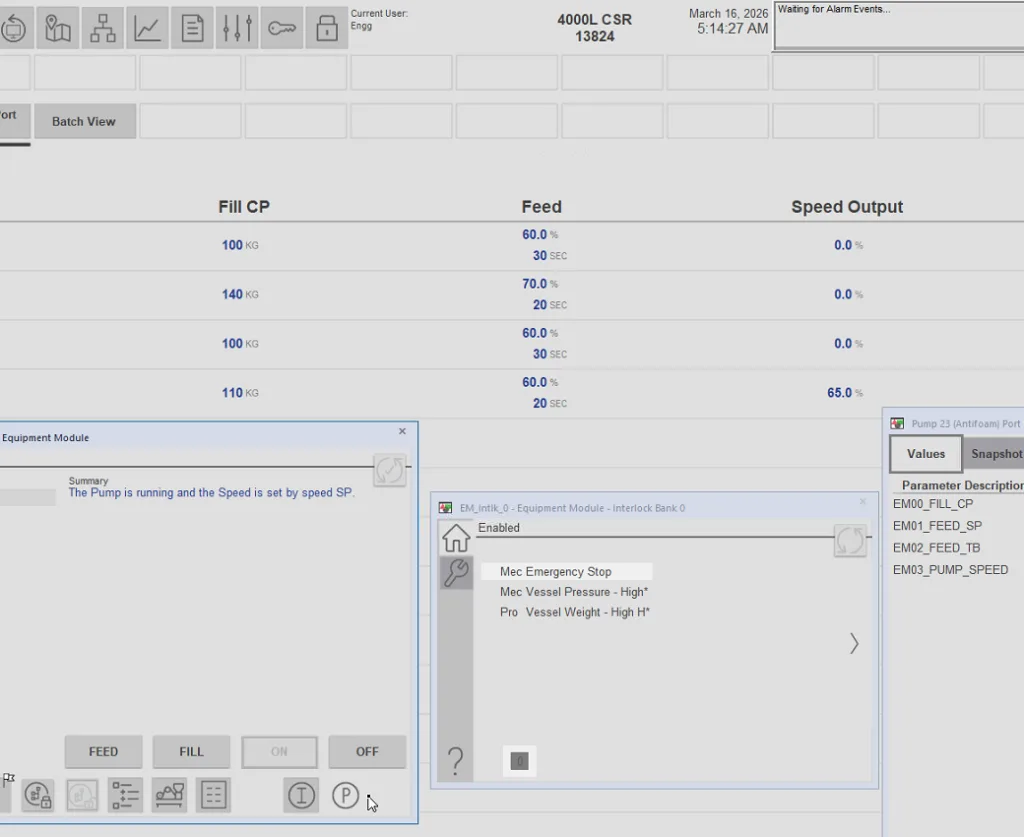
We support Emerson DeltaV, Rockwell PlantPAx, and other supplier platforms, integrating ABEC equipment and third-party systems into a cohesive architecture. Our Synaptic™ HMI approach delivers a consistent operator experience while respecting your site standards.
PROVEN TO DELIVER
THE HIGHEST LEVEL OF CONTROL
In biomanufacturing, small variations can create large consequences. Manual intervention, disconnected platforms, and inconsistent control strategies introduce risk—especially as processes scale, product portfolios expand, and facilities become more complex.
ABEC’s automation specialists design and build integrated control systems that align equipment, data, and workflows. Our open architecture approach makes it easier to standardize, expand, and support your system over time—whether you want to use your preferred platform, open-source components, or custom functionality built from ABEC’s development library.
We apply a structured system lifecycle (SLC) and GMP lifecycle architecture aligned with GAMP 5 and 21 CFR Part 11 expectations—helping mitigate risk from specification through validation, change control, and long-term support.
It’s not automation for automation’s sake. It’s one integrated system that protects performance—and scales with you.
DESIGNED FROM DAY ONE
When automation is architected alongside the equipment and process design (not added later), you reduce handoffs, compress schedules, and improve start-up readiness for both standalone skids and DCS deployments.
CONNECTING THE WHOLE PROCESS
One system works better when it’s integrated with upstream/downstream equipment—including non-ABEC systems. We align interfaces, data flows, and control strategies to reduce integration risk and help your teams troubleshoot faster.

CONSISTENT OPERATOR EXPERIENCE
We support DeltaV, Rockwell PlantPAx, and other platforms while delivering a consistent, usable HMI approach (Synaptic™) that fits your site standards—reducing training time and improving day-to-day operation.

COMPLIANCE BUILT INTO THE LIFECYCLE
Our GMP lifecycle architecture and SLC approach support traceable requirements, risk-based testing, and validation readiness aligned with GAMP 5 and 21 CFR Part 11 expectations—helping reduce rework during qualification and changes.

ONE ARCHITECTURE. ANY PLATFORM.
OPEN, INTEGRATED AUTOMATION
Whether supporting CSR® single-use solutions, stainless-steel systems, Hybrid BioProcessing environments, ATB™ workflows, or Process Sciences-based solutions, ABEC delivers automation that fits your operation: standalone skid controls, area systems, or facility-wide DCS integration. We design for open architecture—so you can use the platform you want—while maintaining a consistent documentation set and support model across your installed base.
By integrating controls with the broader process (inside and outside of ABEC), our approach mitigates risk and helps preserve design intent from engineering through operation.
ENGINEERING: CONTROL SYSTEM DESIGN & INTEGRATION
Control system design, panel build, and integration aligned to your facility standards—supporting standalone skids through DCS connectivity.
CUSTOM FUNCTIONALITY (ABEC DEVELOPMENT LIBRARY)
Reusable, proven modules accelerate delivery while enabling custom workflows, recipes, and integrations tailored to your process.
OPEN ARCHITECTURE
Designed to integrate with third-party skids, utilities, historians, and plant systems—so your automation can evolve without being locked into a single vendor.
PLATFORM FLEXIBILITY (DELTA V, PLANTPAX & MORE)
We work in your preferred environment—including DeltaV and Rockwell PlantPAx—and can incorporate open-source components where appropriate to meet your standards.
GMP LIFECYCLE & SLC SUPPORT
Documentation and execution aligned with the system lifecycle (SLC) and GMP lifecycle expectations (GAMP 5, 21 CFR Part 11), supporting commissioning, qualification, and change control.